 
The above diagram gives us the formula to calculate the formal charge. As we come to the final step, now we are left with the formal charge concept. The skeleton diagram has been sketched, single, double bonds have been drawn and the octet rule has been verified. After we are done with octet fulfillment, we have to check whether we need to build any double or triple bonds. The penultimate step in this process is based on finding out the requirement of multiple bonding. While we are finding out the Lewis Structure, we also need to focus on the octet fulfillment- starting with the electropositive atoms, let us proceed towards octet completion. It states the tendency of an atom inside a molecule to have eight electrons in its outermost or valence shell. The octet rule is basically concerned with the noble gas electronic configuration. Now that we have drawn the molecular skeleton, we need to focus on the next step, i.e. By doing this, we can understand the position of the single bonds more appropriately. Now, let us sketch the skeleton diagram of the molecule. This is considered to be the central atom. Usually, the atom which is the least electronegative has the highest valence and has the highest number of bonding sites. We also need to identify the central atom. The gain of electrons increases the number of negatively charged electrons therefore we use the ‘-’ sign.įor developing the molecular structure, only knowing the number of valence electrons is not enough. To find out the Lewis Structure of any given molecule, the first step is to find out the total valence electron number.Įlectron loss signifies the increase of positive charge hence we use the sign ‘+’. 
Using dots, single and double lines according to the nature of bonds, the Lewis Structure is the first step towards finding out about the properties of a chemical compound, be it hybridization or polarity. If we are interested in having an idea about the internal structure and nature of a given molecule, we need to learn about the sigma pi bond formation and also about lone pairs.Īlso known as electron dot structure, we use Lewis symbols so that we can find out the electronic configuration of the inside atoms of a molecule or ions. 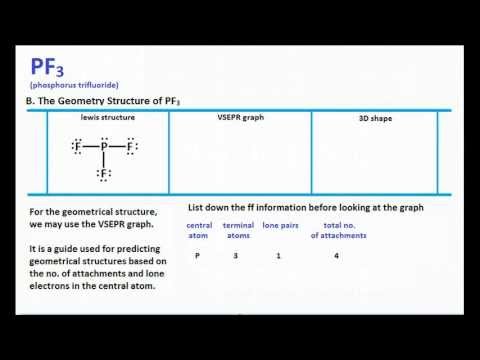
Lewis Structure gives us the diagrammatic sketch of a molecule with details about its chemical bonding nature and electron pair formation. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
